
The average age of diagnosis for children with cancer is 8 years old.

Cancer is the #1 cause of death by disease for children in America.

1 out of 6 children with cancer in America do not survive five years.

Many types of childhood cancer have the same mortality rates that they had decades ago.
It may surprise you to hear that cancer is still killing more children in America than any other disease. Some cancers are still incurable. Even with a more “curable” diagnosis, too many children in the United States don’t have access to the medical care they need. Without treatment, cancer is the same death sentence that it was before modern medicine.

Every day, 41 families learn that their child has cancer.

1 in 264 children will be diagnosed with cancer before their 20th birthday.

Approximately 15,500 children are diagnosed with cancer every year in America.

The average elementary school is attended by at least one child who has or will soon have cancer.
Picture your old elementary school. In all likelihood, at least one of your fellow students was diagnosed with cancer before they could reach their second decade of life. The average student population of an elementary school in America is 473, meaning that most elementary schools are attended by at least one child who has — or will soon have — cancer. In all likelihood, there is a child in your community fighting cancer right now. When you look at the statistics on this page, picture those numbers as grieving parents, siblings who don’t understand what’s happening, and children who can’t visit their friend in the hospital. Picture those numbers as empty chairs in classrooms.
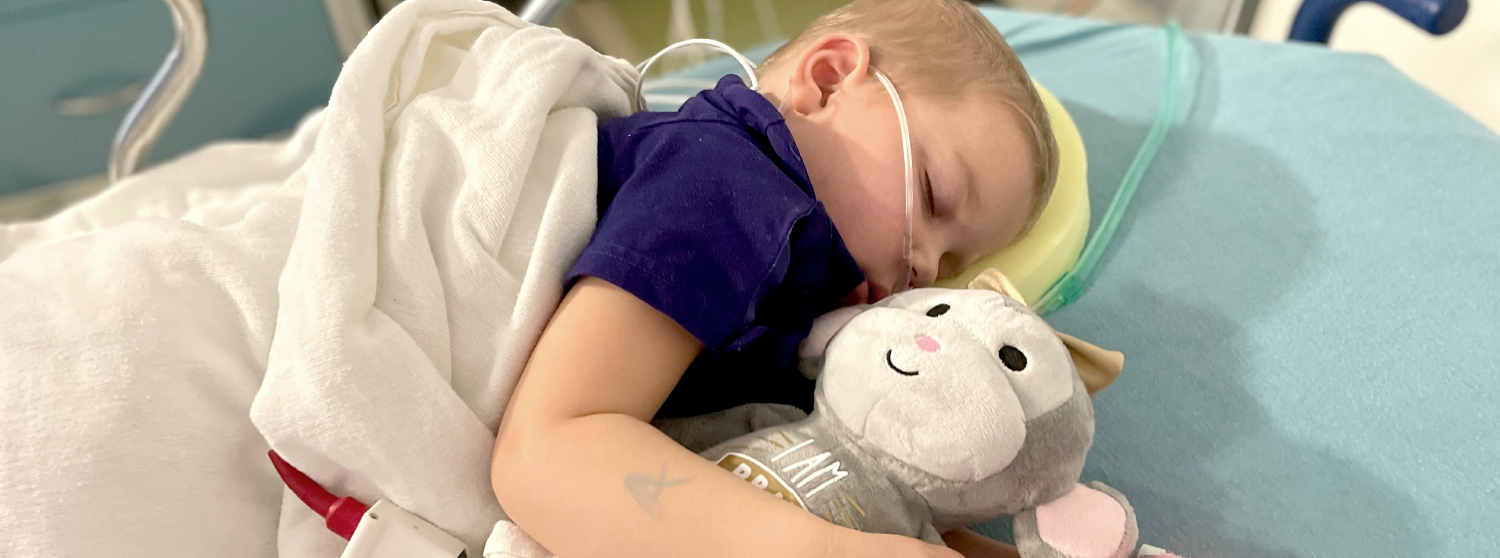
Children today are getting many of the same cancer treatments that children were getting thirty years ago.

Early diagnosis of childhood cancer is often difficult, but late diagnosis can be fatal.

In the past 30 years, 7 drugs have been approved to treat childhood cancer. In this time, over 200 drugs have been approved for adults.*

Studies have shown a correlation between childhood cancer and certain environmental factors like pesticides.
At the American Childhood Cancer Organization, we believe that knowing your enemy is part of fighting it. A critical problem in the fight against childhood cancer has been a lack of coordinated information-gathering that could facilitate more research, enable better treatment options, and empower families and survivors. Therefore, a central element of our mission is helping to ensure that policymakers, researchers, and leaders in the healthcare community, in the United States and abroad, have the tools they need to collect data and share as much information on childhood cancer as possible.

Survivors of childhood cancer are 20 times more likely to experience a major cardiac event before age 50 than their peers.

73% of childhood cancer survivors will suffer a chronic health condition.

42% of survivors will suffer a severe, disabling, or life-threatening condition.

1 in 680 Americans between the ages of 20 to 50 is a childhood cancer survivor.
Cancer will impact a child for the rest of their life. Survivors face a host of health risks and complications, both from the cancer itself and from the toxicity of the treatments. Chemotherapy, radiation, and endless scans can take their toll on a tiny body. Children might lose limbs, be rendered infertile, or suffer longterm cognitive issues. Later in life, they might endure secondary cancers or organ damage. These health conditions can be severe enough to claim the lives of survivors who once fought so hard. And this is just the tip of the iceberg: expensive cancer treatments are not always covered by insurance, leading to devastating financial impacts. The emotional impacts of cancer cannot be overstated either. The isolation and disruption to a normal childhood can have a profound psychosocial impact on children, and the physical pain and existential fear can leave deep scars.
Cancer is a comprehensively traumatizing experience, even for adults. No child is prepared for it. Survival does not always mean recovery.
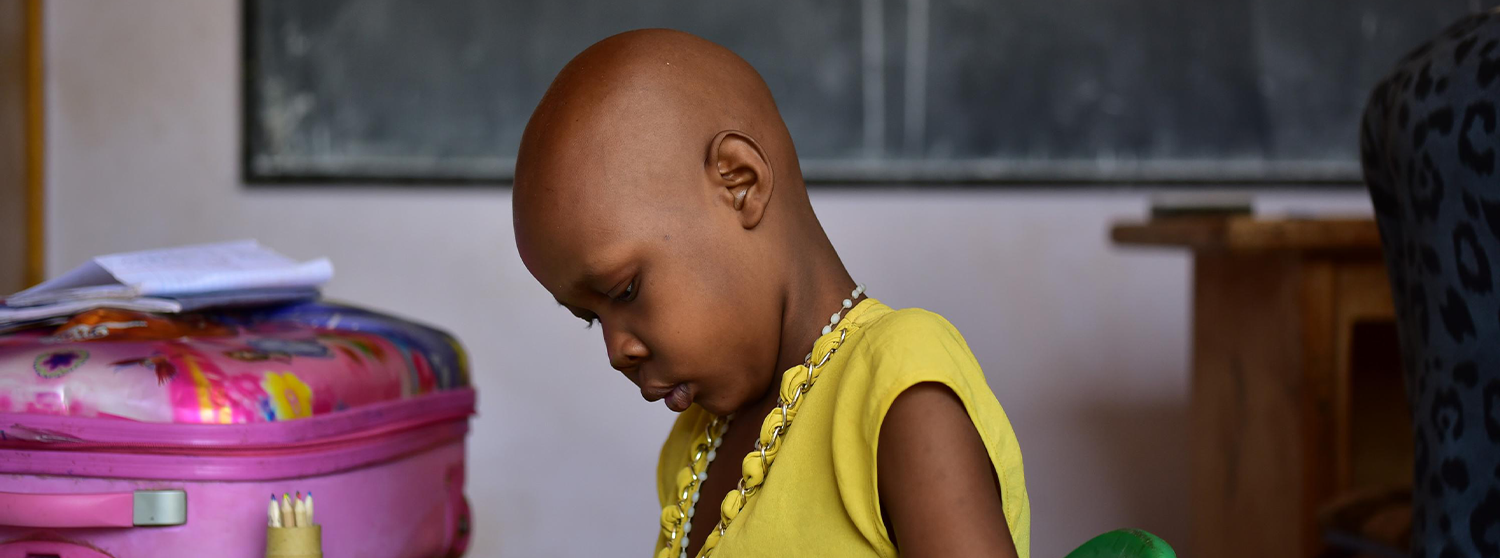
Children with cancer in low-income countries can be up to ten times more likely to die.

Globally, a child is diagnosed with cancer every 80 seconds.

Globally, approximately 413,000 children are diagnosed with cancer each year.

44% of childhood cancer cases worldwide are never diagnosed.
Childhood cancer is a global problem that children and their families struggle with in every corner of the earth. Even with optimal treatment and resources, cancer is a devastating diagnosis. In nations where these resources are limited, the statistics are chilling. Recent studies have highlighted the stark disparity in diagnosis, treatment, and care for children with cancer in different parts of the world. Learn more about the global impact of childhood cancer by clicking here. Learn more about ACCO’s international advocacy work by clicking here.
You can help change these numbers.
Click here to donate.
*There are three pathways for a drug to be approved by the FDA for use in children.
1. The NDA (New Drug Application) is approved first in children
2. The NDA is approved for children and adults at the same time.
3. Approved first in adults, and then in children – this would be a sNDA (Supplemental New Drug Application)
The following 7 cancer drugs approved by the FDA since 1990 to treat childhood cancer, were NDA approvals (4 first in children).
1. Teniposide: NDA approved first in children in 1992 but discontinued after being replaced by etoposide.
2. Clofarabine: NDA approved first in children in 2001.
3. Nelarabine: NDA approved for both children and adults in 2005.
4. Erwinaze: NDA approved for both children and adults in 2011.
5. Unituxin: NDA approved first in children (neuroblastoma) in 2015.
6. Kymriah: NDA approved first in children in 2017.
7. Difluoromethylornithine (DFMO): NDA approved in both children and adults in 2023.


